Call on European governments
MANIFESTO
10 resolutions for a new massive paper-strip test strategy

[ EN ] -
[ FR ] -
[ DE ] -
[ NL ] -
[ IT ] -
[ ES ]
Call on European governments
10 resolutions for a new massive paper-strip test strategy

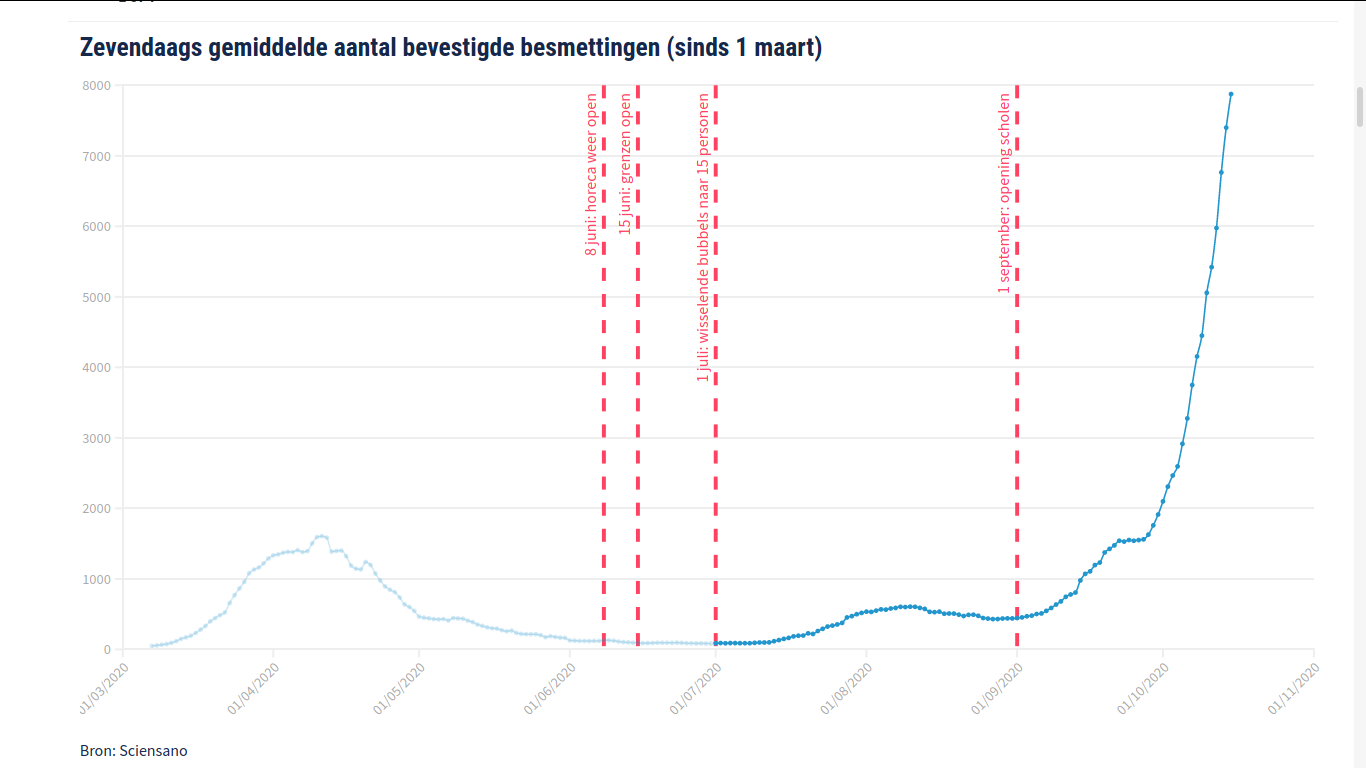
Research shows - and on this topic there is general scientific consensus - that lowcost antigen screening tests can effectively help detect infectious 'corona cases'; and that the best (and most reliable) results are achieved when these rapid tests are applied very regularly (e.g. daily). When used sufficiently frequently by certain population groups, it may even be possible to reduce the number of COVID-19 disease cases in Europe to almost 0 ('close to zero'), because certain members of the statistically most relevant groups (at risk) and the members of these groups that have been recently infected and that should therefore be identified as potential asymptomatic virus spreaders, will immediately discover by way of such high-frequency testing that they are contagious indeed, before they can infect anyone else. This way, the virus is much less likely to spread in our society, while more medical, scientific and diagnostic resources will become available again to effectively combat the pandemic.
Thus, this kind of massive 'population screening', and the so-called 'COVID-19 public health surveillance' screening method that underpins it, not only protects the private interests of each and every individual self-test user, but also - in the first place - it will protect our common interest; starting with the public health of all Europeans.
It is, however, to be expected that the new screening regime such as the one advocated here (based on paper strip saliva tests) will - unfortunately but necessarily - have to be maintained until at least 2023; that is, either until the SARS-CoV-2 virus will have mutated in a positive way and decreased sufficiently in COVID-19 potency (which is unlikely), or until such time anti-viral or disease-modifying agents become sufficiently effective and / or until such time that durable neutralizing or non-neutralizing protective vaccines would be brought onto the market (which may be possible - by 2023 at the earliest), or until such time that all over Europe a minimum group immunity is reached after a sufficiently large cohort of the population has been vaccinated with an effective immune-sterilizing 'SARS-CoV-2 infection-protective' vaccine (which - hopefully by 2023 - is also possible).
Moreover, such expectations are to be compared and contrasted to the objectives of some purely 'COVID-19 disease-protective' emergency vaccines that are now in accelerated development (at the end of 2020), but that unfortunately will offer no or insufficient permanent protection against the viral SARS-CoV-2 infection risks, that are one of the hallmarks of this pandemic. This is even more so because the aforementioned emergency vaccines (planned for 2021 at the earliest) offer an - in any case - uncertain outlook, even if their stated goals and expectations - in any case - are for some very limited levels of efficacy, officially: "the prevention or mitigation of DISEASE, NOT INFECTION", which in terms of effective COVID-19 disease control - according to their manufacturers' own official objectives - amounts to around 70% (and even then: only partial) protection against some major COVID-19 disease symptoms.
In other words, and so there be no misunderstandings on this issue: even if the intended rapid self-tests were to be massively introduced soon, the 'fundamental medical breakthroughs' generally hoped for by public opinion in the field of the viral SARS-CoV-2 transmission and transmissibility mitigation and / or in the field of COVID-19 treatment methods, are only - in the current state of affairs, according to the most optimistic forecasts - expected to materialize in the course of 2022-2023. That is why it will - in any case - always remain necessary, in line with the initial 'flatten the curve' 'infection-mitigation' pandemic strategy, to maintain many of the present prevention policies, in order to first reduce the number of SARS-CoV-2 infections - at all costs - and in order thereafter to mitigate them as much as possible.




And do this: without a doctor's prescription, based on simple antigen paper-strip saliva tests that yield a sufficiently reliable result within 15 minutes (following the example of other paper 'litmus tests' such as the classic do-it-at-home HCG pregnancy urine tests).
In other words: completely change tack and implement a completely new, massive, decentralized screening-test regime, and distribute huge amounts of saliva tests among the population, which on the one hand can be carried out massively and quickly, without any medical intervention, and without any immediate need to call on specialized lab equipment or devices, but which on the other hand reliably demonstrate virus negativity, and thus turn into a 'VIRUS POSITIVE = NOT OK!' or 'NO GO' or 'NOT OK' test result if the user already is, or threatens to become, contagious (or in another rather exceptional situation in case of infrequent testing: if the user may just have been contagious but will certainly soon no longer be - which can then be easily verified by a new test a few hours later).
(1) Immediate self-isolation, until proof to the contrary.
(2) An instant 'confirmatory' paper-strip saliva test (i.e. a confirmatory test based on a different molecular composition, or even based on a slightly different monoclonal Ab technology).
(3) Self-tracing of the user's recent contacts since the last saliva test.
(4) Optionally an additional 'confirmatory' clinical diagnostic (RT-PCR) test.
Particularly appeal to the common sense and civic spirit of the users who carry out these saliva tests in the private sphere of their home, without any obligation to communicate the results to the competent authorities.
For important exceptions to this last principle, see the following Resolution No. 04.


Depending on the concrete circumstances, and only so far as strictly necessary, such 'organized screening' results may be subject to direct, coercive individual or collective measures (such as, for example: isolation or quarantine), but only on condition that the privacy of each individual is respected, including certain basic social, labor law and other personal and/or collective human rights.
To be on the safe side in the context of extensive 'organized saliva tests', supply a number of alternative molecularly deviating 'confirmatory' saliva tests to some of these organizing bodies, which can serve as a double confirmatory test; for example in the event that one of the students, staff, teachers or other members would test positive, or in the event that a whole lot of rapid saliva tests would inexplicably show an exceptionally 'abberrant' number of positive or negative test results. Indeed, the probability of 'false positive' test results can generally be significantly reduced by means of such 'confirmatory' (double confirmation) tests. And in an effort to substantially increase quality control and out of an abundance of vigilance on the part of the 'organizing bodies' organizing and / or coordinating extensive test programs, the use of confirmatory 'validation tests' can also considerably reduce the risk of 'false negative' and/or 'false positive' test results (e.g. by implementing smart 'validation samples'). Therefore: give advance warning and sufficient information to the most obvious organizing bodies about the statistical impact of concepts such as prevalence, sensitivity and specificity on the likelihood of 'false positive' results, and make sure to explain to these 'organizing bodies' how to best monitor and deal with their group test results.
● Paper strip saliva self-tests are relatively less accurate: after all, they are slightly less sensitive and slightly less specific; which can have several consequences.
On the one hand, this can give rise to a greater probability of 'false positive' results,
which can subsequently be compensated for by an almost identical confirmatory saliva test (albeit with a different molecular composition), or by a classic RT-PCR test, or otherwise by simply repeating the same saliva test a few hours later.
On the other hand, there is a limited risk of "false negative" test results - during a short period of a number of hours at the very beginning of the traditional Corona "infectivity peak", which in most cases tends to be asymptomatic; but this limited risk is in turn - certainly if considered across the entire population - offset by factors such as the following:
a) an anyhow relatively low individual "viral load" at the start of infectiousness/transmissibility;
b) a high probability of being 'caught' or 'discovered' in subsequent tests, given the typically-high (e.g. daily) test rate for a representative user of paper-strip self-tests;
c) fast response times in case of the self-tests in comparison with the long turnaround times for highly specific and / or highly sensitive diagnostics where, instead of the hereproposed 15 minutes, the test results will take at least 6 to 24 hours and (all too) often even up to several days, leading to an increase of the actual risk of infection run by a 'positive' patient upon using high-quality albeit 'slow' 'test methods such as RT-PCR laboratory diagnostics, that may eventually turn out to be considerably riskier than in case the same test person - ceteris paribus - would have used a screening method based on the 'rapid' do-it-yourself saliva tests;
d) As a result of the massive deployment of the intended paper-strip saliva tests, many infectious virus carriers are indeed removed from the cohort (ie from the general population), and particularly at those very moments when they are the most contagious (ie during their SARS-CoV-2 infectiousness/trasmissibility peak).
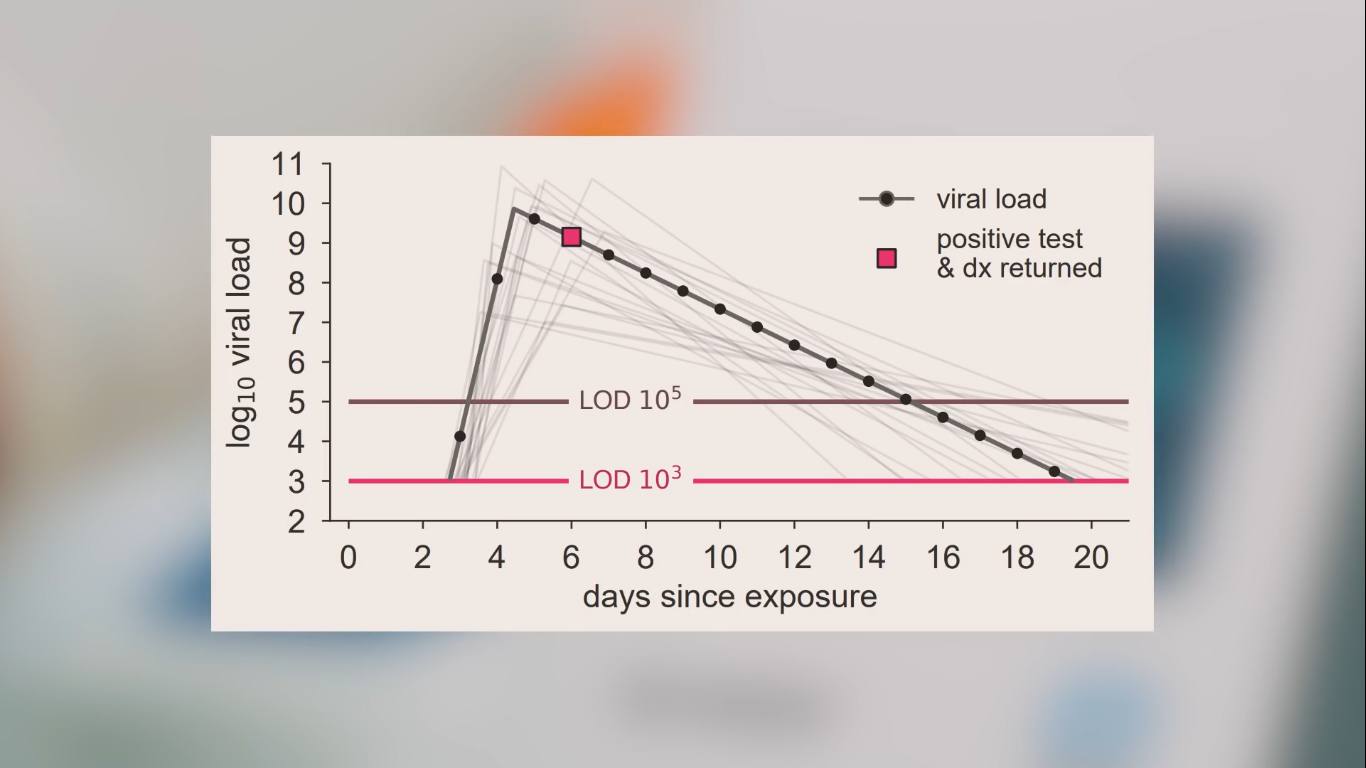
● Paper strip saliva self-tests are considerably more effective: they are much easier to use (at home or outside), and they also provide their users with the intended 'GO - NO GO' test results much faster. This is particularly important when symptomatic or asymptotic Corona virus carriers are going through their viral peak (a period of approximately 60 to 72 hours, with the highest viral load and viral shedding, so with the highest transmissibility, i.e. with the highest risk of infection).
● Paper strip saliva self-tests are considerably cheaper (factor 1/100): after all, they consist (in principle) only of a paper strip test without further medical intervention. Since they are not capital-, labor- or time-intensive (and - at a rate of € 1 per test - are much more cost-effective), antigen saliva paper-strip tests make it possible to test on an individual basis much more frequently: for example daily, or on working days or school days, or prior to boarding an airplane, a bus or a taxi, or for example at very frequent, regular times: every 8, 12, 24 or 48 hours, etc.).
● Paper-strip saliva self-tests are massively 'scalable': unlike other means of testing they can be quickly and easily produced on a massive scale, almost without limits, as they consist of relatively simple paper-strips that are relatively easy to manufacture in specialized printing and packaging factories. Given the ultra-light nature of these strips, their distribution will also be lowcost and predominantly problem-free.







In any case, provide sufficient 'confirmatory tests' (these are almost identical saliva tests, which differ slightly on a molecular level from the standard tests, and therefore can reliably determine whether or not the user is dealing with a 'false positive' result).
For example, with each lot of 100 standard tests, manufacturers could include at the very least 10-15 confirmation tests.
The tenders, specifications and order forms that are drawn up at the time of the purchase should provide for such an arrangement, as this 'second opinion' confirmatory backup system could very well prove very useful later on, to avoid or to solve a range of problems (for example sudden increase in 'false positive' testers) and other related inconveniences, displeasure, lack of confidence, etc. that should best be avoided.
Just as with the distribution of corona face masks, hand outs of saliva tests should be free, following the example of the free distribution of condoms or the free provision of diagnostic tests at the start of HIV-AIDS epidemic in the mid-1980s - a virus for which no effective vaccine has yet been developed (35 years after date).
Note: Despite the fact that the SARS-CoV-2 / COVID-19 pandemic offers significantly better survival prospects in case of infection or disease than the corresponding infection or illness at the start of the HIV / AIDS pandemic, the mantra should still be that prevention is better than cure, so that for the time being the prevention of viral SARS-CoV-2 infections deserves absolute priority, over the search for COVID-19 treatments or vaccines. After all, one cannot simply assume that this ongoing corona crisis situation will definitely be resolved within the next 6 months. And even then..
Moreover, there is an increasing need for a number of humane activities without face masks: certain social and family contacts, group education, sports and cultural events, etc. Such activities can only take place if adequate precautions and preventive measures are taken.
Prioritizing 'frequent and prompt testing' is an essential part of any proper corona infection prevention policy, with the aim of isolating as many infectious individuals as possible, so that normal life can continue and in order for our health facilities not to get 'exponentially' buried under an avalanche of urgent care-intensive COVID-19 cases. The saliva self-test population screening method, as advocated here, is the most effective / efficient (if not the only practically scalable / feasible) strategy to remove from all kinds of communities all over Europe as many, as quickly and as cheaply as possible any infectious cases - everywhere and anywhere in Europe; at least until they test negative again and thus no longer pose any danger to their fellow European citizens.
The inherent strength of the screening method lies in the bio-statistical and epidemiological effects of the continuous, frequent and massive self-testing of individual citizens, who will also immediately discover the outcome of their own test results. It is therefore essential that our fellow European citizens are provided with a massive number of tests, as soon as possible, so that they can start instant-testing themselves in massive numbers.








Repubblica-video-rrtv-650-covid_speranza_100mila_tamponi_a.png)

Markus Lanz vom 13.08.2020-qas8pE5N_iQ-qas8pE5N_iQ.png)


Scientific basis
Adjust the testing and screening policy to the latest scientific knowledge. Let European public health (and scientific insights into this public health) take precedence over the European economy. Take science seriously: including public health economics, public health governance, virology, immunology, epidemics and bio-statistics.
Take the necessary public measures to protect European public health, but do so in the logistically most sensible and statistically most efficient manner; even if less accurate (sub-'Gold standard', sub-RT-PCR) testing means are to be used, such as antigen rapid self-tests.
Encourage the scientific debate, but establish a rapid scientific consensus on the most urgent political challenges that require central control (such as population screening methods based on antigen home tests), and communicate about this with one clear voice.
Thoroughly inform local political and health authorities of all centrally made decisions, as well as of the scientific basis of these decisions.
Subsidiarity
Do on the local level, what can best be arranged and implemented locally. Support local authorities with their policies.
But in the event - as in the case of the corona pandemic - that certain local authorities are in danger of losing control of the situation or that they clearly do not have sufficient command of policy, do not hesitate to intervene, coordinate and organize that policy in their place (and if necessary to also implement this policy in their place) at a higher, more centralized level. In the case of the Europe-wide self-test screening method: at the level of the European Commission, and / or at the national level of the member states.
In case this threatens to degenerate into all kinds of political debates, conduct these discussions with respect for science and by taking into account the policy input and proposals put forward by scientists. That is to say, subject the political decision-making process as much as possible to the established 'applied' scientific consensus.
Enforcement policy
Implement a serious enforcement policy in the context of the corona measures and mandates that have been introduced; especially in the field of the corona face masks and of the corona self-tests. Thoroughly inform local enforcement authorities of all centrally made decisions and of the scientific basis of these decisions. And inform citizens about the medical and scientific consequences of their actions, and about the possible legal consequences of these actions; or of the not performing of such actions.
Reasonable attitudes - Solidarity
As far as can reasonably be expected of European citizens, appeal to their understanding, to their intergenerational solidarity, and to their common sense and moral sense of civic duty.
Anti-Viral Therapeutics & Emergency Vaccines:
Raise awareness and inform the European citizens, and make clear to them that both A) the current development of antivirals, and B) the development / introduction of some new emergency vaccines, will not suffice in the short to medium term to put an end to the high (exponential) risk of infection that is typical for the SARS-CoV-2 virus. Make clear to the European population that the emergency vaccines that are currently being tested are (for the time being) only aimed at a very limited scope of COVID-19 disease control objectives, but that they will certainly not be sufficient to effectively reduce the acute interpersonal SARS-CoV-2 contamination risks (which, for example, emanate from pre- and asymptomatic SARS-CoV-2 virus carriers, and particularly from the infamous Corona-super-spreaders).
Hospital Treatment & Diagnostics:
Raise awareness and inform the European citizens, and make clear to them that the recent progress made in hospital treatment techniques will not suffice indeed, whereas the widespread use of traditional diagnostic testing techniques (such as RT-PCR) for purely technical-logistical reasons is simply not suitable to be expanded 50-fold, or to be transposed from the medical laboratories to the kitchen or to the bathroom, to the station, to the office or to the school campus; let alone that the time frame required for these diagnostic tests could easily be reduced to less than 15 minutes - let alone on the basis of a mere saliva sample. Nevermind the fact that the (overworked) services that run this overburdened RT-PCR test infrastructure, and which are under ever more pressure to reduce turnaround times for these testing platforms, are probably unable to guarantee the duefull protection of the privacy / anonymity of their patient data. Let alone that they could offer such privacy guarantees for the forseeable future. Incidentally, proposals are popping up all over Europe all the time that jeopardize the aforementioned anonymity / privacy of test user data.
Screening & Diagnostics:
Therefore, make clear to the European citizens that they should not be too hopefull or naive in the short to medium term.
The mere "testing, testing, testing" paradigm or the mere approval of an "emergency vaccine" (whatever its ultimate efficacy may be) or the mere introduction of "new treatment techniques" will certainly not suffice to exit the crisis in the short to medium term.
Test Regimen & Pandemic Strategy:
Therefore, make clear to European citizens that they will have to adapt - in any case - to the fact that the recent profound behavioral changes, as adapted since March 2020, will also be necessary in the medium term (i.e. well into 2021 and 2022). And that thus - despite the obstacles described hereabove - the competent authorities will have to switch to a number of alternative strategies and new methods to reduce the acute (often invisible, because asymptomatic) contamination risks that arise in the context of this COVID-19 pandemic as caused by the SARS-CoV-2 virus, in their ongoing effort to mitigate the pandemic.
The most important tool - in addition to the classic face mask - to help European citizens keep up with the expected behavioral change (s) and with this kind of drastic social restrictions, is a new self-test screening regime based on paper-strip saliva tests.
PROS / ADVANTAGES of the New Screening Test Regimen:
-
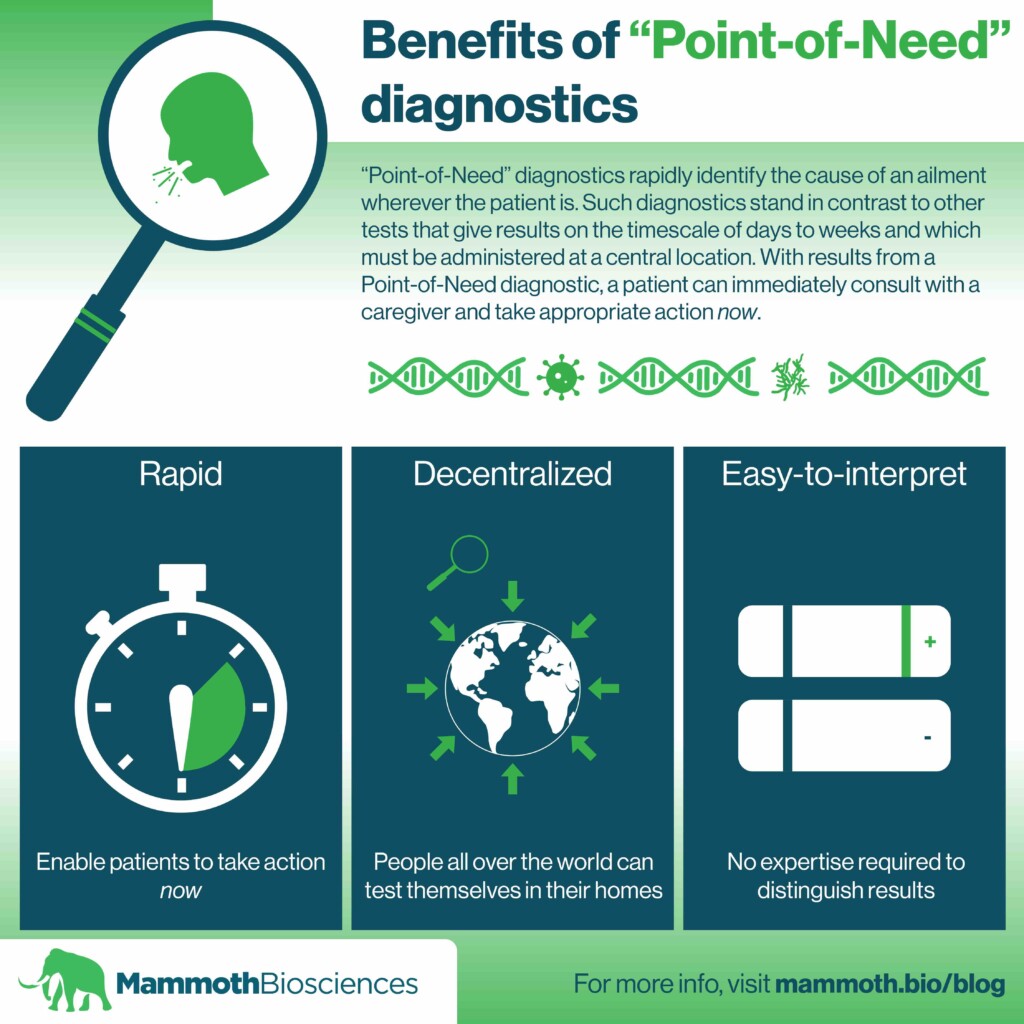
very fast test taking ~ carrying out a saliva test can be done very quickly, when appropriate or convenient (24h/7d)
'point of need' ~ carrying out the tests can be done wherever and whenever it is appropriate ~ unlike traditional (diagnostic) 'point of care' or laboratory tests
routine job ~ daily users can take this test on their own in less than 1 minute on a routine basis, to read the result of the test less than a quarter hour later
ready-to-use ~ new testing regime that can be immediately implemented ~ no need for new anti-viral drugs, no need for new medical treatment methods, no need for additional medically / para-medically trained personnel, no need to wait for the new 'emergency vaccines', no need for doctor visits or COVID-19 hospital admissions, no dire shortages of diagnostic test tube reagents, etc. ~ the necessary technology and infrastructure are already largely available
‘fast positives’ ~ virus positive cases get an almost instantaneous 'NO GO' or 'NOT OK' test result, and can adjust to this result immediately
rapid isolation ~ virus positive testers can go into isolation immediately within 15 minutes after the test is administered, with the possibility of an additional 'confirmatory' saliva test and / or a confirmation based on a gold standard clinical RT-PCR diagnosis
possibility of immediate counter-assessments ~ direct access to a confirmatory test ~ for confirmation purposes, a limited number of identical paper-strip saliva tests are also supplied with every 100 paper strips that work on the basis of an alternative molecular composition (= double-check)
"Fast negatives" ~ virus negative cases get the 'GO' or 'OK' result of their test very quickly, and can continue the activities that are planned for the rest of that day in an unhindered / unabated way; albeit - evidently - without prejudice to the continued observance of the applicable preventive precautions
user-friendly ~ test results are easy (and without risk of confusion or differences of interpretation) for laymen and users to read and to understand
safe ~ by definition the testing protocol involves self-testing ~ in other words, no assistance from third parties or specialized personnel is required, so that these third parties can never become infected during the taking of the test ~ unlike with PCR tests, no "Martians"/PPE are required, which in turn saves a lot of time and money
low cost ~ can be applied on a massive scale by the entire population (eg daily, at less than € 1 per test) ~ economic leverage effect ~ negative opportunity costs + return on investment
"scalable" ~ test that can be applied massively (= by the masses + frequently) in the short to medium term ~ ideal for pandemic screening and / or population screening ~ this is a very interesting feat not just for the users, but also for their organizations and authorities
statistically relevant ~ (structured) test results can assist scientists and policymakers in their decision-making ~ decentralized population screening = a cheap research, development and policy tool
practical / effective triage tool ~ interplay between screening and diagnostics ~ the new paper-strip saliva test screening method is an ideal supplement and / or precursor and / or selection and triage tool for traditional diagnostic tests, which addresses the massive demand / need for RT-PCR tests, whereas at present such massive numbers of RT-PCR-tests can absolutely not be handled by hospitals and diagnostic labs
anonymous ~ (in principle) no need for track & trace ~ protection of privacy ~ protection against 'big brother' and against so-called 'digital surveillance capitalism'
comfortable ~ can easily be taken at home by laymen - no need for terrifying nose swabs, no need for complex lab equipment
compact ~ is portable and stowable ~ can easily be carried in a pocket in a jacket or in a backpack or in a handbag
child-friendly ~ comfortable for children aged 7 to 77, and possibly for those who are younger or older
practical for traveling ~ eg public transportation, airplanes, etc.
practical for on the road ~ eg work, school, hospital, theater, station, airport, stadium, place of worship, workshop, Christmas party at grandma's, etc.
CONS of this new (less accurate) public screening rapid testing regimen:
-
limited risk of "false negatives" if the test is not administered correctly and / or if the test results are not correctly read and / or misinterpreted
~ However: all kinds of precautions can be taken by the user himself/herself (e.g. assistance of children and the elderly, 4-eyes principle within the same family, pointing-and-calling method, test in a quiet room such as a bathroom, etc.). Organizing organizations can also take extra precautions. And in the first place, the manufacturers themselves will of course take the best precautions - as much as possible and as useful as possible. In addition, the government and the media can also raise awareness among the population about the risks of 'false negatives', which will always exist anyway (as they do with other tests), and which of course should not be underestimated. ~ Past experience with other self-tests (such as pregnancy tests, HIV tests, etc.) shows that this type of risk does not have to be an insurmountable problem, and that in developed countries (such as the EU member states) these risks practically can be reduced to almost zero. But even then, even if something goes wrong now and then, the ultimate global effect of this screening method remains predominantly positive, and its ultimate impact remains much better than anything that has been tried so far.
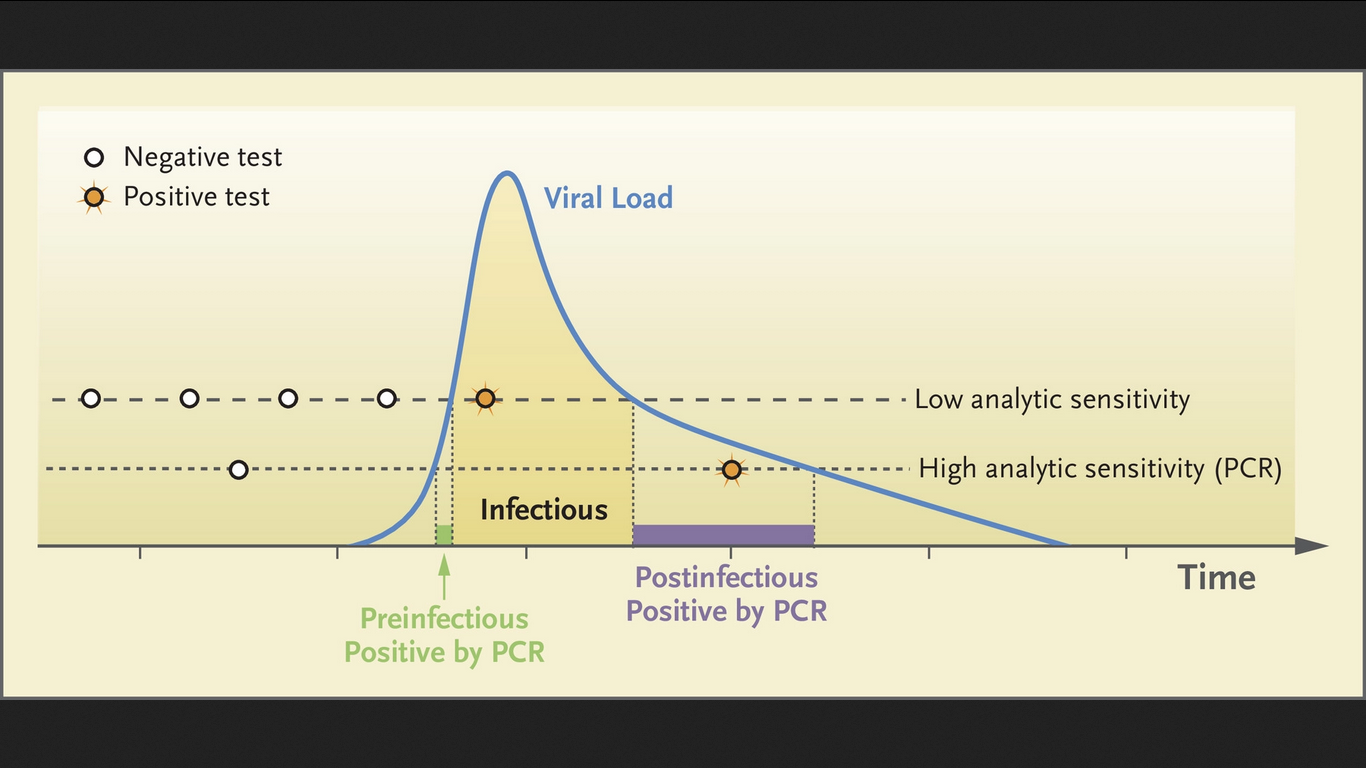
limited risk of 'false negatives' at the (in any case asymptomatic/presymptomatic) very beginning of the 'highly-virus-infectious phase'; this is the 'Virus Infectious / Transmissible Phase with high viral loads and high viral shedding' = 'Ultra-Ansteckende Phase'
~ However: during their so-called 'viral peak' (60-72h with highest risk of infection) this 'initial risk' for false negatives in virus-positive test users is not statistically relevant (thus negligible) from an epidemiological point of view; although it may be useful to remind each test user at an individual level of the existence of the (limited) probability of 'false negatives' at the very beginning of the virus-contagious phase: this risk is not to be 100% neglected indeed, so that other precautions must still be permanently observed.
likelihood of "false positives" (especially given certain typical Bayesian effects), which may give rise to an increasing demand for additional RT-PCR tests, as well as give rise to unnecessary panic, anxiety, work disabilities, school quarantines, etc.
~ However: this risk is largely offset by the additional special 'confirmatory tests' that are included with each batch of standard tests, and that reduce the probability of 'false positive' test results (after a double saliva test) to less than 1/1000 (~ <0.1%).
risk of unexpected escalations and / or other butterfly' or 'bullwhip' effects as a result of some technical details that currently still need to be - at long last - clarified (and preferably as soon as possible), because otherwise they could cause confusion / disinformation with the users of the respective tests, as they will be marketed by different manufacturers. Obviously, what we are dealing with here are simple screening paper-strip tests and not diagnostic devices, but nevertheless there exists a risk for some (admittedly technically-scientifically perfectly explainable) differences in the field of test criteria (specs / specifications) as used by the different Ag saliva test manufacturers; which could indirectly lead to confusion and / or misplaced dissatisfaction among test users, a phenomenon that should therefore be avoided as much as possible.
After all, there is a real possibility of:
(a) divergent test results of scientific samples, (partially due to :)
(b) divergent quantitative and qualitative benchmark and threshold specifications as used by the various saliva test producers. On this very issue, some notable suggestions were launched in recent weeks (among others by certain academic circles in the US and in Berlin), but today the transparency needed to make rapid progress in this field is still lacking.
This concerns, for example, the criteria (to be applied) for 'viral loads & shedding' / 'RT-PCR-ct cycle threshold equivalents'; and this both in terms of the relevant ct values and the VL / ct calibration methods. These are important in delineating what actually constitutes a "positive" and what actually constitutes a "negative" saliva test.
In addition, there is a real possibility that the various saliva test producers apply different criteria with regard to the exact method/protocol to be followed by the individual private users for administering the saliva test; among other things each depending on possibly divergent test specifications (e.g. as a result of differences in the molecular composition of the actual antigenic test strips, which may or may not be open source), in function of diverging views on quality control, in function of user support 'at the point of use', etc.
~
However, this mainly concerns scientific-philosophical discussions. Those can quickly (and easily) find a technical / economic / administrative solution: within the acceptable safety margins and within the probability intervals for screening tests; especially in the framework of the pandemic emergency situation Europe finds itself in. Moreover, the quasi-100% reliability of the saliva tests at the time of a so-called 'viral peak' (i.e. the period of 60-72 hours with the highest risk of infection) must also be considered as a key success driver for the lowcost antigen tests. Hence, possibly divergent criteria and, later on, the risk of divergent tests-results between the different saliva test platforms - in case of a virus-positive test - are actually statistically irrelevant (and therefore practically negligible from an epidemiological point of view); whereby it can not or may not be expected that each test user would (wish to) take these differences into account on his/her individual level. Nevertheless one should caution against a cacophony of differing expert opinions or differences in diagnostic interpretations, which may lead to the test users losing confidence or becoming confused and disinformed. In any case, it would be intellectually dishonest for certain public authorities and / or certain academic bodies and / or certain big-pharma companies to abuse this kind of backbench discussions to block the necessary transition to the new screening test-regime. Such hesitations simply amount to culpable negligence on the part of those responsible. After all, what we are dealing with here are (by definition slightly less accurate) mass public health surveillance screening tests, and not (by definition highly accurate) clinical diagnostic tests. This is precisely the crux of the story, and one should therefore refrain from confusing the population / citizens / test users about the tradeoffs at hand ...
limited risk of technical problems and teething problems, whether or not in combination with incorrect use and / or incorrect interpretation of the confirmatory tests supplied with each batch of standard tests, and whether or not organized by 'test-organizing' organizations. This is all the more so, because the 'viral cell load' of a simple saliva sample can be lower than an equivalent nasopharyngeal swab, possibly leading some to conclude 'that opportunities or signals are being missed'
~ However, these risks can be managed to a significant degree, by way of a phased-in deployment of this new low-tech testing technology, by way of preliminary testing and simulations, by way of sampling and quality control, by way of appropriate training and by way of a Europe-wide awareness-raising campaign that is aimed at the individual testers, at the organizing organizations, as well as at some of the health care personnel. Speed and ease of use are precisely the drivers required for widespread public support for these antigen auto-tests as they will underpin their massive, frequent use, eventhough they are less reliable to begin with. After all: one should not put the cart before the horse; and what clearly prevails here is that the European population continues to frequently test itself in massive numbers, without quitting or giving up because of all kinds of discomforts or inconveniences. What counts is that the decentralized population screening and public health surveillance programs can continue unabated. In other words, and as strange as this may sound: in the case of the modern Ag SARS-CoV-2 saliva tests (and this is particularly true from an epidemiological point of view) the ease of use and the fast turnaround times prevail over the accuracy of the test, which comes in second place. Obviously, one must continue to take as much care as possible (or as useful) to avoid testing incidents and testing accidents, yet especially the speed, the massive numbers and the high frequency, but also the low cost and the comparative ease of use, should prevail over the fact that these Ag saliva tests are somewhat less accurate than the gold standard RT-PCR tests.
limited risk of dangerous behavior and / or a careless attitude in some who think that - in the case of a negative, ie "OK" or "GO" test result - they can let go and start taking unnecessary risks: both in the context of social distancing and personal prevention measures, and in the context of the testing strategy; e.g. in case they no longer regularly observe the frequency and the user instructions such as they apply for the respective saliva test regimens.
~ However, once again, an appropriate enforcement policy, in combination with a Europe-wide awareness-raising campaign, can work miracles, especially among certain population groups (e.g. among children, among the elderly, among tourists, among university students, among the homeless, among refugees, among drug addicts. or also: in the case of schools, associations, airlines, bus companies, organizers of sporting events, etc.). In addition, past experience with other home tests (pregnancy tests, HIV tests, etc.) in developed countries such as the EU member states demonstrates that this kind of risks is certainly manageable, and that it is possible to rely on the common sense and civic spirit of our fellow Europeans.










 - WHO Update (3 August 2020).png)









- -
For instance, an RT-PCR test (total cost: up to 100 €) will sometimes turn out a positive result, up to many weeks after the original symptomatic SARS-CoV-2 virus infection, i.e. at a time when the tested individual has probably not been contagious for weeks, because RT-PCR testing can sometimes still detect 'ineffective' genetic virus RNA fragments, which - given the typical 'gold standard' high specificity and particularly (in casu:) the 'gold standard' high sensitivity of the RT-PCR test - will often lead to a misleading 'positive result', and thus also might lead to all kinds of (misplaced) anxiety, discomfort and inconvenience for the tested person and his/her environment.
example (1):
the case where a former COVID-19 patient relying on the diagnostic RT-PCR test (cost: 100 €) still tests positive 7 weeks after disappearance of the disease symptoms, (long) after having ceased being virus contagious, and thus without being able to transfer the virus to people in his / her environment. In a case like this, the RT-PCR test will give a "false true positive" result (since - in some cases - RNA fragments from a fragmented corona virion can still be detected by the RT-PCR many weeks after the initial COVID-19 disease), while a modern antigen paperstrip saliva test (cost: 1 €) would - ceteris paribus - simply test "truly true negative" for SARS-CoV-2-tranmissibility. Evidently, the latter situation offers a much more useful / soothing answer to the test users concerned - while this antigenic paper strip self-test platform is much cheaper, convenient and faster, to start with.
- -
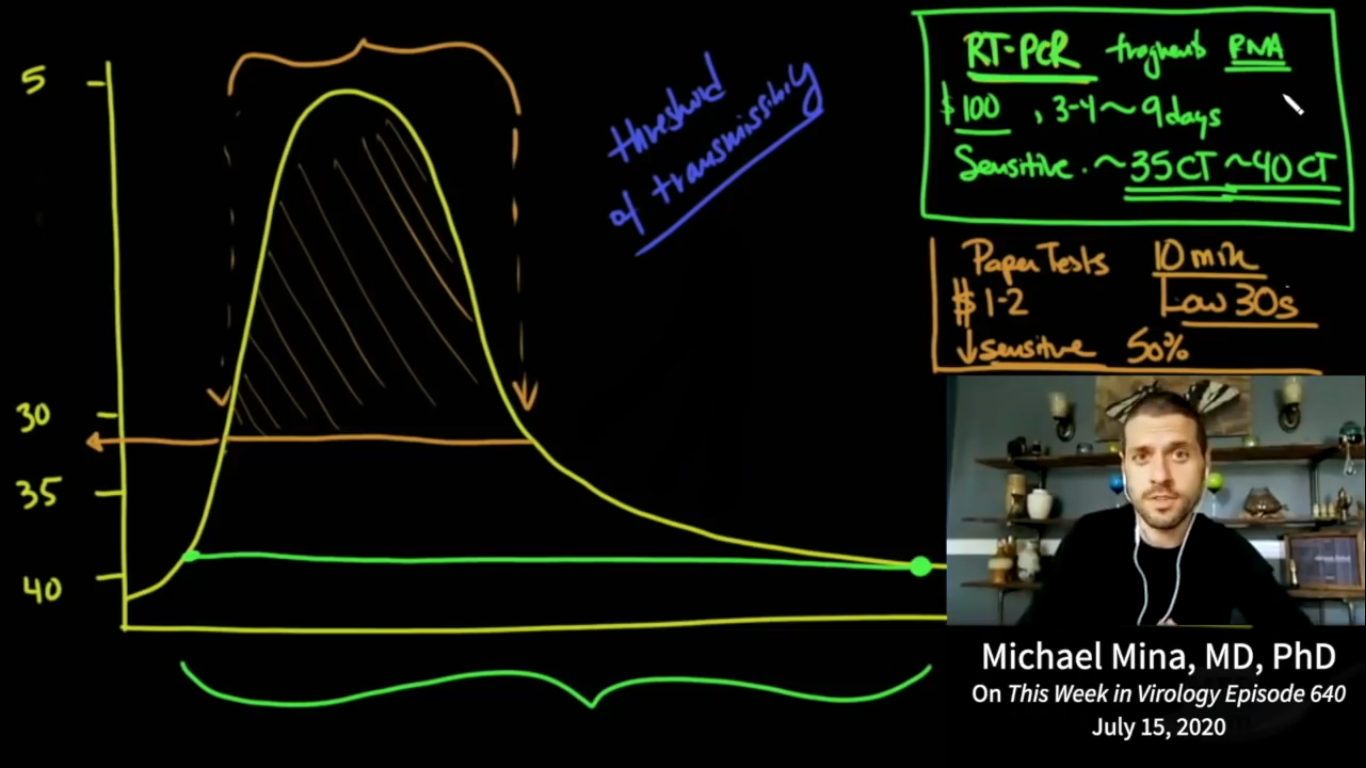
For instance, taking an RT-PCR test (cost: 100 €) can be very time consuming, forcing the test user having to deal with very long waiting lists, queues, testing times, protocols and response times (the so-called total 'turnaround times'), so that it can take an unreasonably long turnaround time before one obtains the test result. Yes, even to the extent that the RT-PCR turnaround times are sometimes so long that the tested individual may long since have been at the origin of further contagion in his/her personal environment. Again, a modern antigenic paper-strip saliva test (cost: 1 €) - ceteris paribus - is likely to give the test-user a compelling "NOT OK" / "NO GO" test result within 15 minutes (not accounting for another 15 minutes for an additional confirmatory test in case the first test is positive indeed), giving the self-test user the opportunity to become immediately aware of the real risk of contamination posed by him/her and of the absolute need for immediate self-isolation. From this point of view, in these concrete circumstances, the antigenic saliva self-test-user and his/her environment are objectively-statistically consisiderably better (safer) off. The availability of much faster (and also much cheaper and easier-to-use) tests, characterised by their almost immediate 'instant' turnaround times, should also allow and motivate large swaths of the population to carry out their own tests massively and frequently (e.g. on a daily basis), thus being one of the most important success factors for this new 'public health' testing strategy. See also Mina et al.: 'Test sensitivity is secondary to frequency and turnaround time for COVID-19 surveillance' (medRxiv preprint doi: https://doi.org/10.1101/2020.06.22.20136309, September 8, 2020).
example (2):
the case of a nasal swab RT-PCR test administered in a hospital: this test is administered by specialized staff in the hospital, before being analysed according to a time-consuming protocol/procedure that is handled with highly specialized equipment operated by highly trained para-medical personnel; with a total reporting and response time back to the tested clinical 'patient' that is all-too-often exceeding 24 hours. Such diagnostic laboratory tests are - in any case - relatively expensive, with an estimated total cost (even without internalizing every external cost) of more than € 100 per test; instead of less than 1 € for a paper strip saliva test.
- -
MACRO-testing ~ 1€ paper strip tests ~ COVID-19-public-health-surveillance ~ 'Quick & Dirty' ~ European population screening ~ the 'SCREENING method':
This public mass screening method mainly focuses on the 'social demonstration' of "Corona test NEGATIVITY": this testing-strategy relies on the individual test users themselves to carry out the tests easily, quickly and cheaply. After all, all is needed is for the individual users to administer the paper-strip tests themselves (cost: approximately € 1 per test). This test method can also be used in an 'organized' way by organizing institutions.
example:
the case of a school: a daily paper strip self-test of all students and of all staff members (estimated cost: less than 1 € per test) can be done before leaving home in the morning, or otherwise immediately upon arrival at school. Indeed, the individual users (or their parents) would each find out the 'GO' / 'NO GO' or 'OK' / 'NOT OK' result on the paper strip within 15 minutes after taking their self-test.
An additional argument for this so-called 'COVID-19 surveillance' method is the private, anonymous nature of the antigenic 'disposable' saliva tests: in principle, each user takes the test on his/her own - at their own initiative and with respect of their personal privacy. In other words, this decentralized anonymous public health 'surveillance' is based on the so-called auto-screening principle (in reality this comes down to an epidemiological population survey). Therefore it should certainly not be confused with other (digitized) forms of 'digital surveillance state' aspects of our modern European healthcare, some of which - as became apparent in the course of the ongoing corona pandemic - are all too often lurking around the corner: telephone appointments, telephone consults (whether or not based on video conference calls), electronic 'track & trace' guest lists, corona apps with geo-location and / or bluetooth recognition, electronic patient files, robotized invoice processing, automatic data exchange between institutions and labs, cloud computing, artificial intelligence, digital outsourcing, data mining, etc. All of which is - by definition - completely out of the question here,.. since everything is still - literally - settled on paper.
- -
MICRO-testing ~ 100€ laboratory tests ~ SARS-CoV-2 detection ~ 'Lean & Clean' ~ useful for scientific or medical research ~ the 'DIAGNOSTIC method':
This private diagnostic laboratory detection method focuses mainly on the medical demonstration of 'Corona test POSITIVITY': one will be able to rely on very precise, very accurate (highly-sensitive / highly-specific) clinical technology that is typically used in hospitals or in clinical laboratories, using capital-, labor- and time-intensive equipment, protocols and reagents (total cost: approx. € 100 per test). This test method is not only expensive, but also inherently slow and cumbersome; and it is therefore difficult to use in an 'organized' way, which means that it will usually be taken on an individual basis.
example:
the case of a so-called 'testing street with Martians in PPE': a professional basketball player came back from vacation 2 weeks ago, participated in a meeting with the coaching staff 1 week ago, and in the meantime found out that some of the fellow guests at his vacation-hotel on their return home were showing COVID-19 symptoms. The basketball player has never fallen ill himself, but the club management wants to make 100% sure (also in order to protect the coaching staff) whether the athlete in the meantime became infected (albeit asymptomatically), and whether the athlete himself could possibly also have posed and/or still poses an infection risk for his immediate enviroment. This can - in this specific case - be verified very precisely by means of an RT-PCR test carried out on all those involved; while the individual 'infectiousness' of each of them could easily be screened for by means of a do-it-yourself saliva test - e.g. in an 'organized' setting: every day in the morning and in the afternoon, at the beginning of each basketball training session.